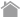SDS-PAGE is an analytical technique to separate proteins based on their molecular weight.
The principle
When proteins are separated by electrophoresis through a gel matrix, smaller proteins migrate faster due to less resistance from the gel matrix. Other influences on the rate of migration through the gel matrix include the structure and charge of the proteins.
In SDS-PAGE, the use of sodium dodecyl sulfate (SDS, also known as sodium lauryl sulfate) and polyacrylamide gel largely eliminates the influence of the structure and charge, and proteins are separated solely based on polypeptide chain length.
SDS is a detergent with a strong protein-denaturing effect and binds to the protein backbone at a constant molar ratio. In the presence of SDS and a reducing agent that cleaves disulfide bonds critical for proper folding, proteins unfold into linear chains with negative charge proportional to the polypeptide chain length.
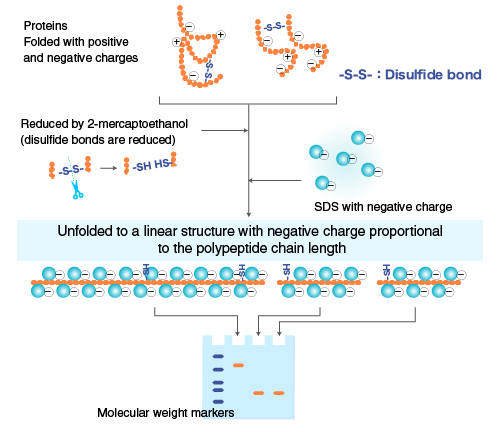
Polymerized acrylamide (polyacrylamide) forms a mesh-like matrix suitable for the separation of proteins of typical size. The strength of the gel allows easy handling. Polyacrylamide gel electrophoresis of SDS-treated proteins allows researchers to separate proteins based on their length in an easy, inexpensive, and relatively accurate manner.
Procedure
| Step-by-step procedure | |
|---|---|
| Gather combs, glass plates, spacer (silicone tubing), and binder clips. A comb is used to make wells (lanes) to load samples. Use an appropriate comb depending on the sample size. Example: Use an 8-lane comb for 7 samples and molecular weight markers. 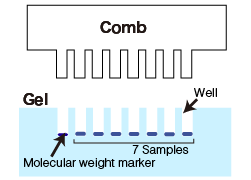 |
 |
| Thoroughly clean the glass plates with ethanol, and assemble the gel casting mold. |  |
Pour acrylamide solution for a separating gel. Overlay with water to prevent contact with air (oxygen), which inhibits polymerization. Allow acrylamide to polymerize for 20-30 minutes to form a gel. Remove the overlaid water. Proteins migrate at different rate depending on the concentration of the separating gel. Use an appropriate gel concentration for your target protein. Using a higher acrylamide concentration produces a gel with a smaller mesh size suitable for the separation of small proteins. In general, an acrylamide concentration between 6 and 15% is used. Gels with an acrylamide concentration gradient (gradient gels) are also used. |
 |
| Pour acrylamide solution for a stacking gel; insert a comb and allow the acrylamide to polymerize. Proteins are highly concentrated when they migrate through a stacking gel prior to entering a separating gel. The concentration occurs due to the difference in the rate of migration of glycine ion, chloride ion, and proteins, as illustrated below. 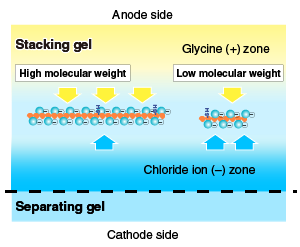 |
 |
![]()
| Step-by-step procedure | |
|---|---|
| Add sample buffer to samples, and mix by flicking the tube. Heat the samples at 100°C for 3 minutes in a heat block. Centrifuge at 15,000 rpm for 1 minute at 4°C, and use the supernatant for SDS-PAGE. |
 |
![]()
| Step-by-step procedure | |
|---|---|
| Remove the binder clips, spacer, and comb from the gel assembly, and mount the gel in the electrophoresis apparatus using binder clips. Pour running buffer into the upper and lower chambers of the electrophoresis apparatus, and remove air bubbles and small pieces of gel from the wells and under the gel using a syringe. |
 |
| Load samples and molecular weight markers in wells. |  |
Turn on the power supply, and run the gel until the dye (BPB) in the sample buffer reaches the bottom of the gel.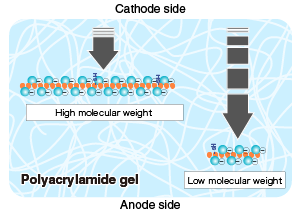 |

|
| Remove the gel assembly from the electrophoresis apparatus. Remove the gel from the glass plates using a spatula, and prepare for subsequent analysis. | 
|