Deacetylase Assay Kits
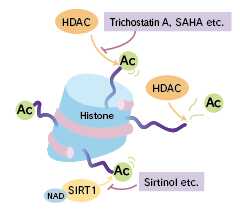 Histone deacetylase (HDAC) is considered to play a crucial role in regulating gene expression by changing nucleosome structure. HDAC is also thought to participate in regulation of cell cycle and differentiation, and it has been reported that the failure of this regulation leads to some types of cancer.
Inhibition of HDAC activity by HDAC inhibitors such as trichostatin A (TSA) and suberoylanilide hydroxamic acid (SAHA) induce differentiation and/or apoptosis of transformed cells in vitro and inhibit tumor growth in a mouse model. It has been reported that HDAC inhibitors are effective for the medical treatment of acute promyelocytic leukemia (APL) and various cancers. Thus, HDAC inhibitors are expected to function as new anti-tumor drugs and antibacterial reagents. It is thought that screening of histone deacetylase inhibitors is likely to be further carried out, as one way to discover additional substances with similar properties.
Histone deacetylase (HDAC) is considered to play a crucial role in regulating gene expression by changing nucleosome structure. HDAC is also thought to participate in regulation of cell cycle and differentiation, and it has been reported that the failure of this regulation leads to some types of cancer.
Inhibition of HDAC activity by HDAC inhibitors such as trichostatin A (TSA) and suberoylanilide hydroxamic acid (SAHA) induce differentiation and/or apoptosis of transformed cells in vitro and inhibit tumor growth in a mouse model. It has been reported that HDAC inhibitors are effective for the medical treatment of acute promyelocytic leukemia (APL) and various cancers. Thus, HDAC inhibitors are expected to function as new anti-tumor drugs and antibacterial reagents. It is thought that screening of histone deacetylase inhibitors is likely to be further carried out, as one way to discover additional substances with similar properties.
HDACs Deacetylase Fluorometric Assay Kit
CycLex HDACs Deacetylase Fluorometric Assay Kit measures the activity of HDAC by the basic principle of changing an HDAC reaction into the activity of the protease. Since it is very simple to measure common protease activity and it can be performed at a low price, the measurement of HDAC activity in most laboratories is possible if they are equipped with a fluorescent reader for microtiter plates. Considering that the use of fully automatic apparatus to measure fluorescence intensity has become widespread, HDAC activity measurement, which could not be made by the conventional method, is now possible with the CycLex HDACs Deacetylase Fluorometric Assay Kit using the same equipment. This new method of measurement should dramatically raise the efficiency of inhibitor screening and biochemical analysis of these enzymes.
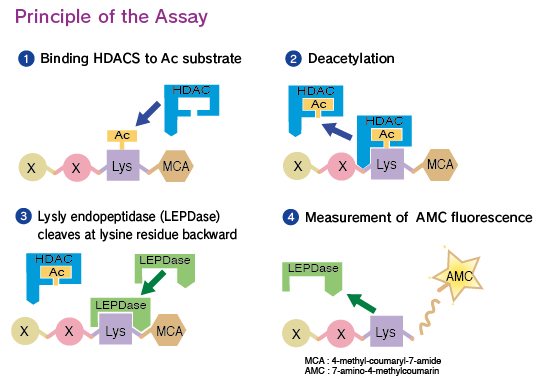
This measuring principle and kit are covered under CycLex's patents.
U.S. Patent No. 7,033,778 and No. 7256013
European Patent No. 1243658
Japanese Patent No. 4267043
Canadian Patent No. 2392711
 HDACs Deacetylase Fluorometric Assay Kit Ver.2
HDACs Deacetylase Fluorometric Assay Kit Ver.2
HDAC8 Deacetylase Fluorometric Assay Kit
The human HDACs are organized into three different classes based on their similarity to yeast HDAC proteins. Class I enzymes are ubiquitously expressed and include HDAC1, -2, -3, and -8, which are homologous to the yeast RPD3 protein. Class II includes HDAC4, -5, -6, -7, -9, and -10, which are similar to yeast HDA1 and are expressed in a tissue-specific manner. The Sir2-like class III HDACs, including SIRT1 to -7, require NAD+ for enzymatic activity.
It has been reported that HDAC8 is important for the growth of human tumor cell lines and has a
distinct inhibition pattern that differs from that of HDAC1 and -3, which both share 43% sequence identity with HDAC8. These findings lead to open the way to the development of selective inhibitors of this subtype as potential novel anticancer therapeutics.
 HDAC8 Deacetylase Fluorometric Assay Kit Ver.2
HDAC8 Deacetylase Fluorometric Assay Kit Ver.2
SIRT Deacetylase Fluorometric Assay Kit
Sir2 is a conserved protein and was recently shown to regulate lifespan extension both in budding yeast and nematode. In 2000, it was reported that the yeast Sir2 protein is a NAD+-dependent histone deacetylase that plays a critical role in transcriptional silencing, genome stability and longevity. In mammals, the homologs of Sir2 have been named sirtuins (SIRT), with seven members in a family termed SIRT1 through SIRT7. They share a conserved central deacetylase domain but have different Nand C termini and display distinct subcellular localization, suggesting different biological functions.
SIRT1 was shown to regulate the activity of the p53 tumor suppressor and inhibits apoptosis. These results have significant implications regarding an important role for SIRT1 in modulating the sensitivity of cells in p53-dependent apoptotic response and the possible effect in cancer therapy. Since the function of p53 is made to strengthen powerfully by using together with DNA damaging reagent, it is expected that inhibitor of SIRT1 becomes an effective anticancer drug.
 SIRT1/Sir2 Deacetylase Fluorometric Assay Kit Ver.2
SIRT1/Sir2 Deacetylase Fluorometric Assay Kit Ver.2
 SIRT2 Deacetylase Fluorometric Assay Kit Ver.2
SIRT2 Deacetylase Fluorometric Assay Kit Ver.2
 SIRT3 Deacetylase Fluorometric Assay Kit Ver.2
SIRT3 Deacetylase Fluorometric Assay Kit Ver.2
 SIRT6 Deacetylase Fluorometric Assay Kit Ver.2
SIRT6 Deacetylase Fluorometric Assay Kit Ver.2



 Deacetylase Assay Kits
Deacetylase Assay Kits