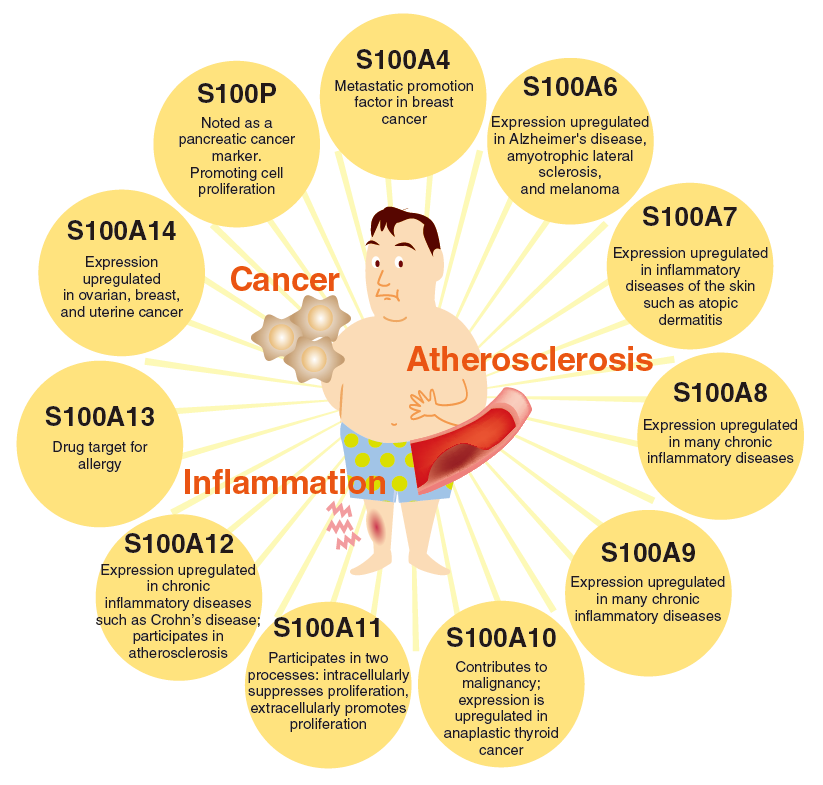
S100 Protein family ELISA Kits
S100 protein family of calcium binding proteins with 2 distinct helix-loop-helix motifs (EF-hands) shows cell typespecific expression and consists of 20 members. The protein family functions by being secreted extracellularly as well as involved in intracellular signaling. S100 protein family has various, complicated functions, many of which remain unknown. Recently, S100 protein family was also reported to function as ligands of RAGE. The CircuLex S100 Protein Detection kit series are designed to measure the concentration of S100 protein family from serum, plasma and other biological media.
S100A4 ELISA Kit
Members of the S100 protein family are low molecular mass acidic proteins characterized by cell-type-specific expression and the presence of 2 EF-hand calcium-binding domains with different affinities for calcium. Elevated levels of one of the S100 protein, S100A4, are closely associated with the process of metastasis in breast and other cancer cells in rodent animal models and in human cancer specimens. S100A4 or its mRNA is found at an elevated level in metastatic relative to nonmetastatic rat and mouse tumor cell lines and malignant relative to benign human breast tumors. Elevation of the level of rat or human S100A4 in benign rat mammary tumor cells results in the acquisition of metastatic capability by some of the cells. In transgenic mouse models of breast cancer, elevated levels of S100A4 in neu oncogene-induced, or in murine mammary tumor virus-induced, benign mammary tumors yield lung metastases. In colorectal adenocarcinoma specimens, elevated levels of immunocytochemically detected S100A4 are associated with the more malignant carcinomatous regions of the primary tumors and with liver metastases.
S100A6 ELISA Kit
S100A6 also known as calcyclin, a small acidic protein of which molecular weighs is about 10.5 kDa, belongs to the S100 calcium-binding protein family. These family members share a common S100 calcium-binding motif and are involved in several regulatory functions that include protein phosphorylation, some enzyme activities, the dynamics of cytoskeletal components, transcription factors, and Ca2+ homeostasis, and also cell proliferation and differentiation. S100A6 is most abundantly expressed in fibroblasts and epithelial cells and has also been found in some neurons, astrocytes, smooth muscle cells, cardiac myocytes, platelets and lymphocytes. S100A6 is also expressed by osteoblasts and up-regulated markedly during osteoblast differentiation; however, its roles in bone physiology are unknown. In addition, S100A6 is overexpressed in several tumor cells with high metastatic activity, e.g. melanoma. Therefore S100A6 may also be involved in tumorigenesis. The general function of S100A6 remains unclear, but evidence suggests that it is involved in cell cycle regulation and exocytosis, and in the regulation of heat shock proteins and cytoskeletal dynamics. Interestingly overexpression of S100A6 has also been observed in patients suffering from Alzheimer disease or amyotrophic lateral sclerosis.
S100A7/Psoriasin ELISA Kit
Human Psoriasin was first identified as an over-expressed secreted protein in psoriatic skin. Psoriasin also called S100A7, is distributed in the cytoplasm of keratinocytes in normal human epidermis and is present at the cell periphery in terminally differentiated keratinocyte. The peripheral distribution observed in differentiated cells may be important, since, under some conditions, S100A7 may be released from keratinocytes. Indeed, S100A7 has been shown to function as a chemotactic agent and as a cytokine and to attract CD4+ lymphocytes and neutrophils. S100A7 overexpression is seen in
many epidermal inflammatory diseases, including atopic dermatitis, mycosis fungoides, Darier's disease, and inflammatory lichen sclerosus at atrophicus. S100A7 expression is also increased in invasive skin cancers such as squamous cell carcinoma, and squamous carcinoma in situ, but not in basal cell carcinoma.
Moreover, concentration of S100A7 is reported to be elevated in serum and cerebrospinal fluid of patients withAlzheimer's disease. S100A7 is attracting attention as a biomarker of Alzheimer's disease.
S100A8 ELISA Kit
S100A8/MRP8 and its hetero-dimerization partner S100A9/MRP14 belong to the low molecular weight calcium-binding protein, S100 family, which are composed of two EF-hands flanked by hydrophobic regions at either terminus and separated by a central hinge region.
In human, 100A8 is usually co-expressed with S100A9. Both proteins are expressed during myeloid differentiation, are abundant in granulocytes and monocytes, and form heterodimeric complexes. In human, they have been associated with several inflammatory diseases: elevated serum levels of S100A8 and S100A9 have been found in patients suffering from a number of inflammatory disorders including giant cell arteritis, cystic fibrosis, rheumatoid arthritis, dermatoses, chronic inflammatory bowel disease, chronic bronchitis, some malignancies and autoimmune diseases.
Both proteins are localized predominantly in the cytoplasm. An increase in the intracellular calcium concentration leads to a translocation of the proteins from the cytoplasm to the plasma membrane. In addition, it was demonstrated with human monocytes that both proteins are secreted by an energy-consuming pathway.
S100A9 ELISA Kit
The S100A9/MRP14 protein and S100A8/MRP8 belong to the low molecular mass calcium-binding S100 proteins, they are composed of two distinct helix-loop-helix motifs (EF-hands) flanked by hydrophobic regions at either terminus and separated by a central hinge region. In human, S100A9/MRP14 is usually co-expressed with S100A8/MRP8. Both proteins are expressed during myeloid differentiation, are abundant in granulocytes and monocytes, and form heterodimeric complexes. Although a number of possible functions for S100A8-A9 heterocomplex, including antimicrobial activity, have been proposed, the exact role of these proteins in cell metabolism is still unclear. In human, they have been associated with several inflammatory diseases: phagocytes expressing S100A9 belong to the early infiltrating cells and dominate acute inflammatory lesions; in addition, elevated serum levels of S100A8 and S100A9 have been found in patients suffering from a number of inflammatory disorders including cell arteritis, cystic fibrosis, rheumatoid arthritis, dermatoses, chronic inflammatory bowel disease, chronic bronchitis, some malignancies and autoimmune diseases. Both proteins are localized predominantly in the cytoplasm. An increase in the intracellular calcium concentration leads to a translocation of S100A9 from the cytoplasm to the plasma membrane. In addition, it could be demonstrated with human monocytes that both proteins are secreted by an energy-consuming pathway, which is dependent on an intact microtubule network and involves protein kinase C. It was reported that S100A9 is a molecular target of quinoline-3-carboxamides, which are currently in Phase II and III development for treatment of autoimmune/inflammatory diseases in humans.
S100A10 ELISA Kit
S100A10, a member of the S100 family of Ca2+-binding proteins, is a dimeric protein composed of
two 11-kDa subunits. The protein is cytosolic when present as a dimer. Typically, S100A10 is found in
most cells bound to annexin A2 as the heterotetrameric (S100A10)2-(annexin A2)2 complex, AIIt, in a
calcium-independent manner. The formation of AIIt results in the translocation of S100A10 to the
plasma membrane. S100A10 has been shown to regulate plasma membrane ion channels as
well as cytosolic phospholipase A2 In addition to an intracellular distribution, it has also been
established that the heterotetrameric form of S100A10 is present on the extracellular surface of many
cells. Extracellularly, the S100A10 subunit functions as a plasminogen receptor. The
penultimate and ultimate carboxyl-terminal lysines of this subunit bind tPA and plasminogen and
regulate the stimulation of tPA-dependent plasminogen activation.
Although S100A10 is expressed fairly ubiquitously in most cells and tissues, high levels of both p11
and A2 were found in 100% of anaplastic thyroid carcinomas, and correlated with their aggressive
behavior. In a comprehensive study of S100 gene expression in over 300 primary breast cancers,
both p11 (S100A10) and S100A11 were selectively upregulated in basal versus nonbasal breast cancer
subtypes, but did not predict overall survival.
S100A11 ELISA Kit
S100A11 is expressed ubiquitously in various tissues at different levels, with high expression level in the skin. In the cultured skin keratinocytes, S100A11 is localized in cytoplasm of resting cells and relocated to the cell periphery following calcium treatment.
S100A11 was shown to bind to annexin A1 and the S100A11/annexin A1 complex is a heterotetramer consisting of two S100A11 and two annexin A1 proteins. Ca2+ binding to S100A11 induces a conformational change that exposes a hydrophobic surface for interaction with target proteins. In addition to binding to annexin A1, S100A11 has been shown to interact with annexin A6, actin and transglutaminase, and is capable of forming a heterodimer with S100B through subunit exchange.
S100A12/EN-RAGE ELISA Kit ver.2
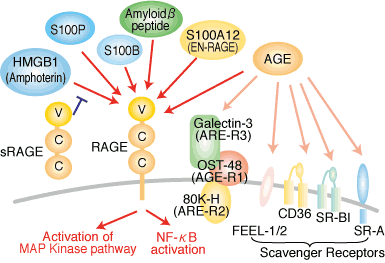 S100A12, also referred to as EN-RAGE (Extracellular Newly identified RAGE-binding protein), was first reported as a ligand of RAGE (Receptor for Advanced Glycosylation End products). Interaction of EN-RAGE (S100A12) with cellular RAGE on endothelium, mononuclear phagocytes, and lymphocytes triggered cellular activation, with generation of key proinflammatory mediators.
S100A12, also referred to as EN-RAGE (Extracellular Newly identified RAGE-binding protein), was first reported as a ligand of RAGE (Receptor for Advanced Glycosylation End products). Interaction of EN-RAGE (S100A12) with cellular RAGE on endothelium, mononuclear phagocytes, and lymphocytes triggered cellular activation, with generation of key proinflammatory mediators.
S100A12 serum concentrations indicate neutrophil activation in JRA, cystic fibrosis, Kawasaki disease and Crohn's disease.Its function as a proinflammatory factor secreted by activated neutrophils makes this protein a potential target for future therapies.
 S100A12/EN-RAGE ELISA Kit ver.2
S100A12/EN-RAGE ELISA Kit ver.2
S100A13 ELISA Kit
S100A13, a small acidic protein that molecular weighs about 10-11 kDa, belongs to the S100 calcium-binding protein family. S100A13 seems to function in exocytosis, since it is one of the targets of two antiallergic drugs, amlexanox and cromolyn, which inhibit degranulation of mast cells.
Recently, S100A13 was shown to be associated with the fibroblast growth factor 1 (FGF-1)/p40 synaptotagmin-1 (p40Syn-1) complex, and amlexanox is able to repress the release of S100A13 from the complex. These findings suggest that S100A13 might be involved in the regulation of FGF-1 and p40Syn-1 release in response to heat shock. Another possibility might be that S100A13 is secreted together with the FGF-1/p40Syn-1 aggregate.
S100A14 ELISA Kit
S100A14, a protein identified from a human lung cancer cell line subtractive cDNA library, encodes a deduced 104-amino acids protein with 2 EF-hand Ca2+-binding domains, a myristoylation motif, a glycosylation site, and several potential protein kinase phosphorylation sites.
It has been demonstrated that S100A14 transcript is down-regulated in many immortalized and tumor cell lines from different tissues. In contrast, studies on human primary tumors, including lung and breast, revealed predominant up-regulation of the gene at the mRNA and protein level.
S100A14 shows heterogenic expression in tumors, demonstrating its overexpression in ovary, breast, and uterus tumors and under-expression in kidney, rectum, and colon tumors, a pattern suggesting distinct regulation with potentially important functions in malignant transformation.
S100P ELISA Kit
One of the least studied members of the S100 family is S100P, a 95-amino acid protein first purified from placenta with a restricted cellular distribution. Expression of S100P has been noted in esophageal epithelial cells during their differentiation. S100P expression has been also noted in various cancer cell lines including breast cancer and colon cancer. S100P has also been reported to be involved in immortalization of cells by activation of Erks and NF-κB via RAGE.



 PCSK9 ELISA and PCSK9-LDLR in vitro Binding Assay Kits
PCSK9 ELISA and PCSK9-LDLR in vitro Binding Assay Kits