Glycated Proteins/AGEs related Kits
AGE ELISA Kit
 Reducing sugars react with protein amino groups to form a diverse group of protein-bound moieties with fluorescent and cross-linking properties. These compounds, called advanced glycosylation end products (AGEs), have been implicated in the structural and functional alterations of proteins that occur during aging and long-term diabetes.
Reducing sugars react with protein amino groups to form a diverse group of protein-bound moieties with fluorescent and cross-linking properties. These compounds, called advanced glycosylation end products (AGEs), have been implicated in the structural and functional alterations of proteins that occur during aging and long-term diabetes.
Nε-(Carboxymethyl)lysine (CML) is one of the AGEs and its concentration is increased in patients who have diabetes with complications, including nephropathy, retinopathy, and atherosclerosis. CML is also recognized by receptor for AGE (RAGE), and CML-RAGE interaction activates cell signaling pathways such as NF-κB and enhances the expression of vascular cell adhesion molecule-1 in human umbilical vein endothelial cells.
 CML/Nε-(Carboxymethyl)lysine ELISA Kit
CML/Nε-(Carboxymethyl)lysine ELISA Kit
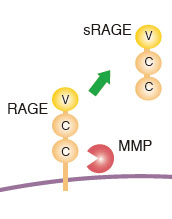
sRAGE ELISA Kit and AGE-RAGE in vitro Binding Assay Kit
 RAGE is a multi-ligand member of the immunoglobulin superfamily of cell surface molecules that is expressed in a variety of cell lines. RAGE is thought to be involved in pathogenesis of various conditions including inflammation, diabetes, Alzheimer's disease, systemic amyloidosis, and tumor.
RAGE is a multi-ligand member of the immunoglobulin superfamily of cell surface molecules that is expressed in a variety of cell lines. RAGE is thought to be involved in pathogenesis of various conditions including inflammation, diabetes, Alzheimer's disease, systemic amyloidosis, and tumor.
Soluble version of RAGE, termed soluble RAGE (sRAGE), created by proteolytical cleavage by matrix metalloproteases can be detected in sera. In addition to sRAGE, another soluble RAGE, as endogenous secretory RAGE (esRAGE) derives from alternative splicing of the RAGE mRNA was discovered. Because of the possible neutralization effect of sRAGE, studies have examined the significance of sRAGE serum concentration in patients with various pathological conditions.
Decreased level of sRAGE is a biomarker for deficient and/or altered inflammatory control in humans. It was shown that reduced level of sRAGE is associated with higher risk of coronary disease. In Alzheimer's disease, there is a decrease in serum sRAGE in comparison with patients with vascular dementia and controls. On the other hand, an increased level of serum sRAGE was found in patients with end-stage renal disease and acute lung injury.
 Human sRAGE ELISA Kit
Human sRAGE ELISA Kit
 AGE-RAGE in vitro Binding Assay Kit
AGE-RAGE in vitro Binding Assay Kit
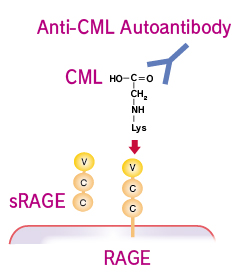
Anti-CML Autoantibody ELISA Kit
Although several AGE structures have been reported, it was demonstrated that Ne-(Carboxymethyl)lysine (CML) is a major antigenic AGE structure. It has been postulated that AGE structures present in vivo could serve as an immunological epitope to raise autoantibodies against AGE structures, particularly CML. It has been reported that the presence of autoantibodies against AGE structures, particularly those against CML adduct in streptozotocin (STZ)-induced diabetic rats and patients with several diseases. The autoantibody against CML adduct was higher in patients with renal failure than in normal subjects or diabetic patients without renal failure. These results suggest that autoantibody against CML might play a possible role in the development of diabetic nephropathy or chronic renal failure.
 Anti-CML mouse autoantibody ELISA Kit
Anti-CML mouse autoantibody ELISA Kit
Glycated Proteins / Advanced glycation end products (AGEs)
Advanced glycosylation end products (AGEs) are formed during the late stage of Mailard reaction, and have been implicated in the structural and functional alterations of proteins that occur during aging and long-term diabetes. Although several AGE structures have been reported, Nε-(Carboxymethyl)lysine (CML) and Nε-(Carboxyethyl)lysine (CEL) are two major stable, nonenzymatic chemical modifications of protein lysine residues resulting from glycation and oxidation reactions.
 Advanced glycation end products (AGEs)
Advanced glycation end products (AGEs)



 PCSK9 ELISA and PCSK9-LDLR in vitro Binding Assay Kits
PCSK9 ELISA and PCSK9-LDLR in vitro Binding Assay Kits