Adipokine/Lipid metabolism ELISA Kits
Adiponectin ELISA Kit
 A reduction in adiponectin serum levels is accompanied by insulin resistance states, such as obesity and type 2 diabetes mellitus. Increased adiponectin levels are associated with type 1
diabetes mellitus, anorexia nervosa and chronic renal failure. Adiponectin concentrations correlate
negatively with glucose, insulin, triglyceride concentrations and body mass index and positively with high-density lipoprotein-cholesterol levels and insulin-stimulated glucose disposal.
A reduction in adiponectin serum levels is accompanied by insulin resistance states, such as obesity and type 2 diabetes mellitus. Increased adiponectin levels are associated with type 1
diabetes mellitus, anorexia nervosa and chronic renal failure. Adiponectin concentrations correlate
negatively with glucose, insulin, triglyceride concentrations and body mass index and positively with high-density lipoprotein-cholesterol levels and insulin-stimulated glucose disposal.
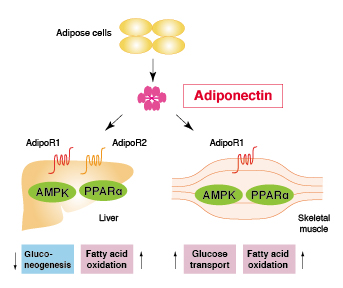
Among two Adiponectin receptors AdipoR1 and AdipoR2, AdipoR2 is highly expressed in the liver. Through the two receptors, Adiponectin activates AMP-activated protein kinase (AMPK) and Peroxisome proliferator-activated receptor α (PPARα), which results in increase of fatty acid oxidation, glucose uptake and insulin sensitivity. Therefore, Adiponectin is regarded as a candidate for treatment of metabolic syndrome.
 Rat Adiponectin ELISA Kit
Rat Adiponectin ELISA Kit
 Human Adiponectin ELISA Kit
Human Adiponectin ELISA Kit
 Mouse Adiponectin ELISA Kit
Mouse Adiponectin ELISA Kit
 Dog Adiponectin ELISA Kit
Dog Adiponectin ELISA Kit
Soluble LOX-1/OLR1 ELISA Kit
Lectin-like oxidized LDL receptor-1 (LOX-1), a 52-kD type II transmembrane receptor for oxidized low-density lipoproteins (ox-LDL) belonging to the C-type lectin family is present primarily on endothelial cells. Accumulating evidences indicate that oxLDL uptake through this receptor induces endothelial dysfunction. This receptor is expressed in the aortas of hypertensive, diabetic, and hyperlipidemic animals and is upregulated in early human atherosclerotic lesions. These results suggest that LOX-1 may be expressed locally and play important roles in atherogenesis by internalizing and degrading oxLDL and in inflammatory responses in vivo. LOX-1 can be cleaved from the cell surface and released as soluble LOX-1 (sLOX-1), and elevated sLOX-1 levels may be indicative of atherosclerotic plaque instability.
 Human soluble LOX-1/OLR1 ELISA Kit
Human soluble LOX-1/OLR1 ELISA Kit
RBP4 ELISA Kit
Members of the lipocalin family, RBP4, FABP4 and NGAL (Lipocalin 2), which are produced from adipocytes, have recently been reported to play important roles in regulating systemic energy homeostasis, insulin sensitivity, and inflammation in animal models. Transgenic overexpression of RBP4 or injection of recombinant RBP4 in normal mice causes insulin resistance. Conversely, genetic deletion of RBP4 enhances insulin sensitivity. it was reported that serum RBP4 levels correlated with the magnitude of insulin resistance in human subjects with obesity, impaired glucose tolerance, or type
2 diabetes and in non-obese, non-diabetic subjects with a strong family history of type 2 diabetes. Elevated serum RBP4 was associated with components of the metabolic syndrome, including increased body mass index (BMI), waist-to-hip ratio, serum triglyceride levels, and systolic blood pressure and decreased high-density lipoprotein cholesterol levels. Adipocyte GLUT4 protein and serum RBP4 levels were inversely correlated.
Therefore, it is suggested that measurement of serum RBP4 levels is useful tool for the identification of insulin resistance before the development of diabetes and cardiovascular risk factors in subjects with varied clinical presentations.
NAMPT/PBEF/Visfatin ELISA Kit and NAPT colorimetric Assay Kit
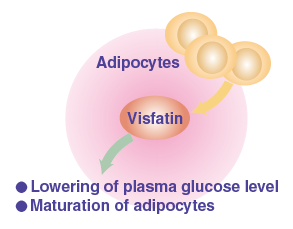 Human obesity-related diabetes and the accompanying metabolic disorders have been specifically linked to increased visceral adipose tissue mass. Visfatin was identified as an adipokine predominantly expressed in and secreted from visceral adipose tissue in both humans and mice. This adipokine is also known as a growth factor for early B-cells called pre-B-cell colony-enhancing factor (PBEF) or nicotinamide phosphoribosyltransferase (NAMPT).
Human obesity-related diabetes and the accompanying metabolic disorders have been specifically linked to increased visceral adipose tissue mass. Visfatin was identified as an adipokine predominantly expressed in and secreted from visceral adipose tissue in both humans and mice. This adipokine is also known as a growth factor for early B-cells called pre-B-cell colony-enhancing factor (PBEF) or nicotinamide phosphoribosyltransferase (NAMPT).
It has been reported that despite visfatin does not include a known secretory signal, some visfatin is secreted from cell by inflammatory stimuli and functions as inflammatory cytokine itself.
FK866, the specific visfatin inhibitor, has been reported to cause NAD+ depletion and induce apoptosis in various cancer cell lines, and is now developed as anti-cancer drug.
The visfatin inhibitor can also be a new candidate of antiinflammatory drug, since visfatin is reported to have proinflammatory effect.
 NAMPT Colorimetric Assay Kit Ver.2
NAMPT Colorimetric Assay Kit Ver.2
 Human NAMPT/PBEF ELISA Kit
Human NAMPT/PBEF ELISA Kit
AIM/CD5L/Spα ELISA Kit
 AIM, also called CD5L/SPα, is a soluble 38 or 40-kDa glycoprotein expressed by macrophages present in lymphoid tissues (spleen, lymph node, thymus, and bone marrow). It belongs to the group B of the scavenger receptor cysteine-rich (SRCR) superfamily that includes the lymphocyte surface receptors CD5 and CD6 among others. AIM is able to bind to not only myelomonocytic and lymphoid cells, but also the surface of several Grampositive and Gram-negative bacterial strains, which suggests that it may play
an important role in the regulation of the innate and adaptive immune systems.
AIM, also called CD5L/SPα, is a soluble 38 or 40-kDa glycoprotein expressed by macrophages present in lymphoid tissues (spleen, lymph node, thymus, and bone marrow). It belongs to the group B of the scavenger receptor cysteine-rich (SRCR) superfamily that includes the lymphocyte surface receptors CD5 and CD6 among others. AIM is able to bind to not only myelomonocytic and lymphoid cells, but also the surface of several Grampositive and Gram-negative bacterial strains, which suggests that it may play
an important role in the regulation of the innate and adaptive immune systems.
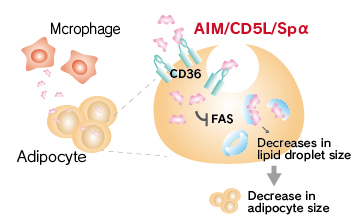
It has been reported that AIM is incorporated into adipocytes via CD36 and inhibits the activity of fatty acid synthase (FAS), which results in decrease in lipid droplet size and adipocyte size.
 Human AIM/CD5L/Spα ELISA Kit
Human AIM/CD5L/Spα ELISA Kit
 Mouse AIM/CD5L/Spα ELISA Kit
Mouse AIM/CD5L/Spα ELISA Kit
FABP4/A-FABP ELISA Kit
 Fatty acid binding protein 4 (FABP4), also designated aP2 and Adipocyte-specific fatty acid-binding protein (A-FABP), belongs to the fatty acid-binding protein super family whose members have relative molecular masses of –15, 000, and it is exclusively expressed in differentiated adipocytes. FABP4/A-FABP is a predominant cytosolic protein of mature adipocytes, accounting for –6 % of total cellular proteins.
Fatty acid binding protein 4 (FABP4), also designated aP2 and Adipocyte-specific fatty acid-binding protein (A-FABP), belongs to the fatty acid-binding protein super family whose members have relative molecular masses of –15, 000, and it is exclusively expressed in differentiated adipocytes. FABP4/A-FABP is a predominant cytosolic protein of mature adipocytes, accounting for –6 % of total cellular proteins.
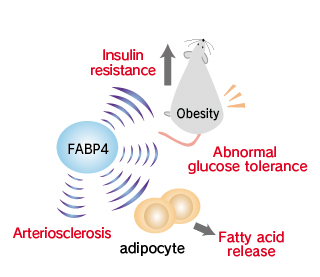
This protein may be an important regulator of systemic insulin sensitivity and lipid and glucose metabolism. Mice deficient in FABP4/A-FABP are protected from development of hyperinsulinemia, hyperglycemia, and insulin resistance in the context of both dietary and genetic obesity. Adipocytes obtained from FABP4/A-FABP -null mice had markedly reduced efficiency of lipolysis in vivo and in vitro and exhibited a 2- to 3-fold decrease in fatty acid release, suggesting that FABP4/A-FABP mediates efflux of fatty acids in normal physiology. In apolipoprotein E-deficient mice, ablation of the FABP4/A-FABP gene conferred remarkable protection against atherosclerosis, which commonly occurs in this rat strain.
By integrating metabolic and inflammatory pathways, FABP4/A-FABP provides a key link between various components of metabolic syndrome.
 Rat FABP4/A-FABP ELISA Kit
Rat FABP4/A-FABP ELISA Kit
 Mouse FABP4/A-FABP ELISA Kit
Mouse FABP4/A-FABP ELISA Kit
FABP5/E-FABP/mal1 ELISA Kit
Human epidermal fatty acid binding protein (E-FABP, FABP5 and in mice called also mal1) belongs to the intracellular FABP family and is expressed in differentiated adipocytes, macrophages, skin, brain, mammary glands and so on. Mouse FABP5/E-FABP/mal1 has a high degree of homology to aP2 (mouse FABP4) and is also an important player in obesity-related disorders. FABP5/E-FABP/mal1 knockout mice exhibited enhanced insulin-stimulated glucose uptake and increased systemic insulin sensitivity, while transgenic overexpression of FABP5/E-FABP/mal1 aggravated insulin resistance and hyperglycaemia. Conversely, a marked compensatory up-regulation of adipocyte FABP5/E-FABP/mal1 expression was observed in FABP4/ aP2-deficient mice.
 Mouse FABP5/E-FABP/mal1 ELISA Kit
Mouse FABP5/E-FABP/mal1 ELISA Kit
 Human FABP5/E-FABP ELISA Kit
Human FABP5/E-FABP ELISA Kit



 PCSK9 ELISA and PCSK9-LDLR in vitro Binding Assay Kits
PCSK9 ELISA and PCSK9-LDLR in vitro Binding Assay Kits