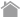Human HLA class I Tetramer
High Specificity of T-Select HLA class I Tetramers
 The T cell surface CD8 enhances T cell antigen recognition by binding to HLA class I molecules. Therefore, we produced T-Select HLA class I Tetramers with one point mutation (Ala245Val) at the HLA α3 domain known to reduce the CD8-HLA interaction. These mutated tetramers showed a greatly diminished nonspecific binding but retained specific binding2). Alterations of CD8 binding by mutation of the HLA greatly improved the specificity of HLA-peptide multimers, thus providing efficient tools to sort specific human T cells for immunotherapy.
The T cell surface CD8 enhances T cell antigen recognition by binding to HLA class I molecules. Therefore, we produced T-Select HLA class I Tetramers with one point mutation (Ala245Val) at the HLA α3 domain known to reduce the CD8-HLA interaction. These mutated tetramers showed a greatly diminished nonspecific binding but retained specific binding2). Alterations of CD8 binding by mutation of the HLA greatly improved the specificity of HLA-peptide multimers, thus providing efficient tools to sort specific human T cells for immunotherapy.
*Please note that α3 mutation is adopted only in Human HLA Class I Tetramers, but not in other species including Mouse MHC Class I Tetramers.
References:
1) Gao GF, et al., Nature 387: 630−634 (1997)
2) Bodinier M, et al., Nat. Med. 6: 707−710 (2000)
The example of staining 1: HLA-A*02:01 CMV pp65 Tetramer
CMV-Positive Sample:
T-Select HLA Class I Tetramers show substantially reduced non-specific CD8 binding (C) compared to Native (wild type) Tetramers (A).
CMV-Negative Sample:
T-Select HLA Class I Tetramers produce accurate results (D) compared to Native Tetramers that produce false-positive results (B).
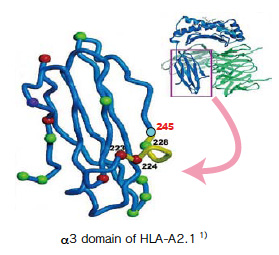
The example of staining 2: HLA-A*24:02 EBV BRLF1 Tetramer
PBMC were separated from peripheral blood of HLA-A*24:02 positive healthy subjects. EBV BRLF1-specific CTLs were induced by the MLPC method and stained with PE-labeled HLA-A*24:02 Negative (Code No. TS-M007-1), or EBV BRLF1 Tetramer with (Code No. TS-M002-1) or without the α3 mutation. Non-specific staining of CD8 positive cells was observed in the sample stained with theconventional, but not the α3-mutated, tetramer.
※This data was analyzed within the lymphocyte gate and 7-AAD negative gate.
Tips for better staining
・ How to use?
・ Staining method
・ Induction of specific CTL
・ FAQ